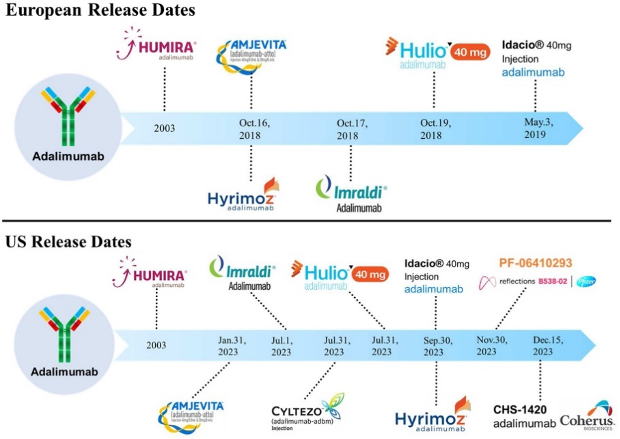
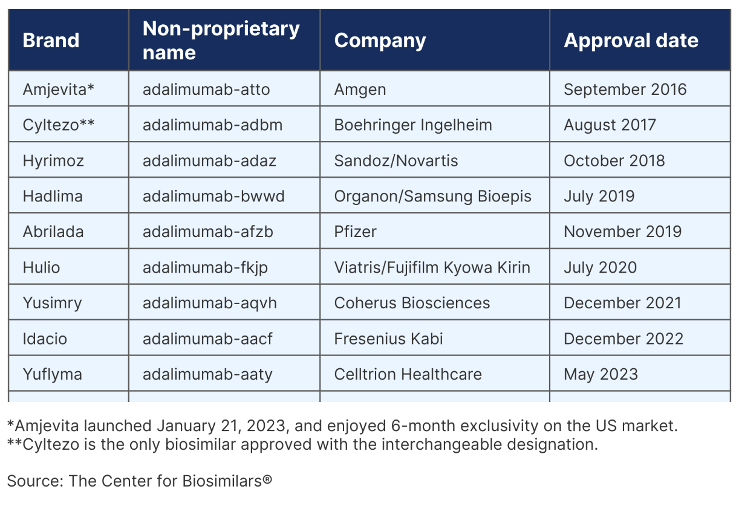
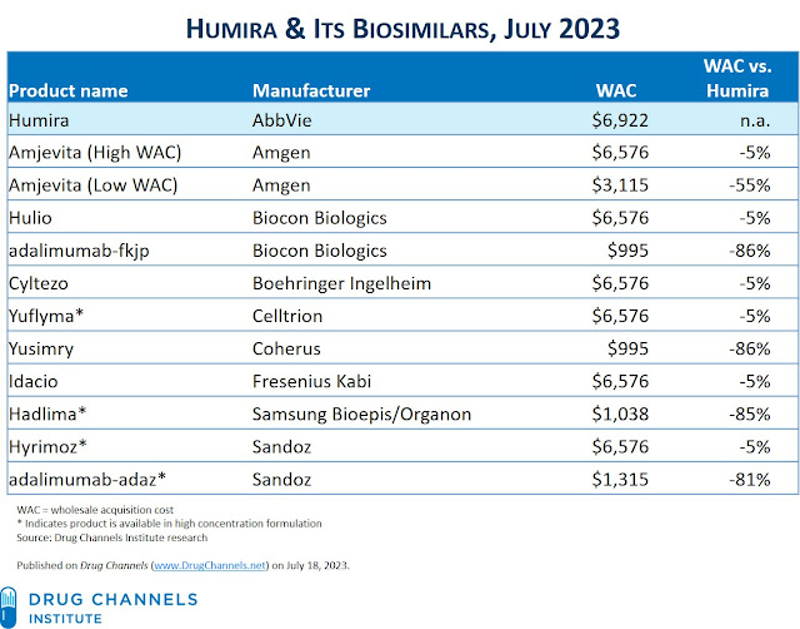
How to Counter Drug Price Increases by Taking Advantage of Personal Importation and other Solutions
After wrapping up the 2023 enrollment, employers are now planning their next moves to mitigate the continuously growing healthcare costs impacting their plans. This is particularly true for pharmacy costs.